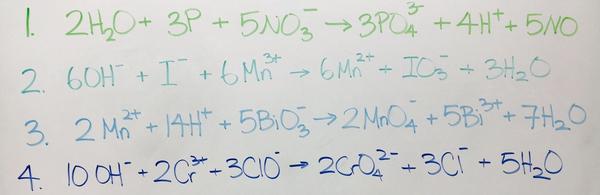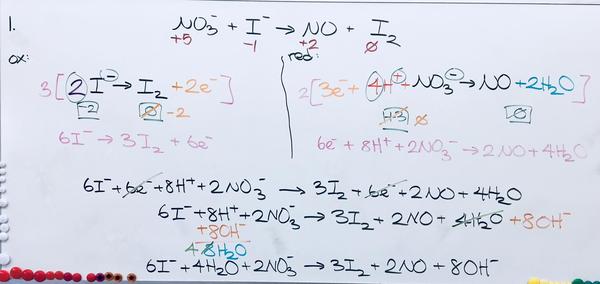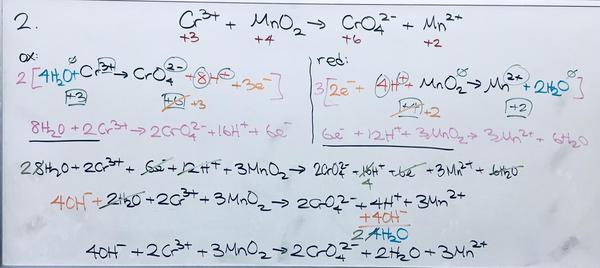2017-2018 Chemistry 1-2 Assignments
- Instructors
- Terms
- Fall 2017
- Spring 2018
- Department
- Science
- Description
-
Files
Upcoming Assignments

No upcoming assignments.
Past Assignments
Due:
Assignment
Check! Semester 2 Final Review
5/19/17 Final Review Pt. 2
62. 170 ft3
63. 279 mL
64. 19.0 atm
65. 310 K
67. 0.242 g Ne
68. 7 L
69.1.88 x 103 L
70. 7.9 x 105 lb H2
71. 395 mL H2
74. 4.4 x 104 cal or 183 kJ
75. dispersion forces; hydrogen bonds & dispersion forces; dipole-dipole interactions & dispersion
76. CS2; HF; CH3CH2OH
78. CS2
79. CH3OH
80. NaCl
82. 2Na + 2H2O --> 2NaOH + H2
Na2O + H2O --> 2NaOH
SO3 + H2O --> H2SO4
2H2O --> 2H2 + O2
83. 2H2 + O2 --> 2H2O
2C4H10 + 13O2 --> 8CO2 + 10H2O
H2C2O4 + 2LiOH --> 2H2O + Li2C2O4
84. Na2O2•8H2O
85. increase P & decrease T
86. surface area, temperature, and stirring
87. CH3OH - polar
89. 28.6% glucose; 2.22 molal
90. 1.00 M
91. 6.94 g CaCl2
92. 0.30 M
93. 0.033 L or 33 mL
94. -8.44ºC
95. (KBP for water is 0.512 ºC/m ) 102.79ºC (ionic - breaks into two)
96. 53.4 g/mol
99. 0.2639 M H2SO4
101. HCO3- = base
H2PO4- = acid
HPO42- = conjugate base
H2CO3 = conjugate acid
| [H+] | [OH-] | pH | pOH | A/B |
| 2.3 x 10-4 M | 4.3 x 10-11 M | 3.64 | 10.36 | acid |
| 2.7 x 10-9 M | 3.7 x 10-6 M | 8.57 | 5.43 | base |
103. HCl; KOH; NaNO3
104. H+ + OH- --> H2O
H3PO4 + 3OH- --> 3H2O + PO43-
2Ag+ + CO32- --> Ag2CO3
106. Rate = 0.213 s-1 [A]1[B]0
109. 9.6
110. right; left; none; right; right
111. right; left; none; left
112. 2.51
113. 1.6 x 10-11 M; 1.4 x 10-2 M
114. H2S < S8 < S2O32- < SO2 < H2SO4
115.
| Ox | Red | Ox ag | Red ag |
| Mg | O | O2 | Mg |
| Al | H | H+ | Al |
| Mn | O | H2O2 | Mn2+ |
116. 2Al + 3Br2 --> 2Al3+ + 6Br-
14H+ + Cr2O72- + 6Cl- --> 3Cl2 + 2Cr3+ + 7H2O
2OH- + Mn2+ + H2O2 --> MnO2 + 2H2O
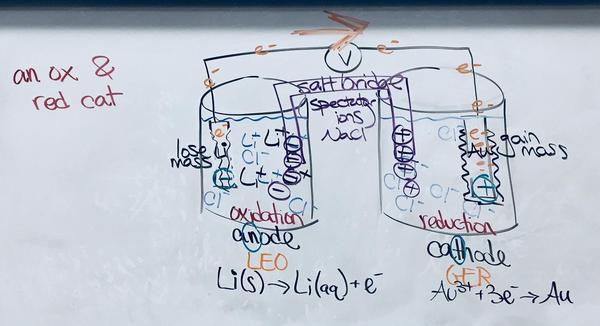
117. anode: Cu; cathode: Rh
e- flow from anode to cathode
cations to the cathode compartment
anions to the anode compartment
Eºcell = 0.28 V
If it helps any, here are some things we memorized during Semester 2:
n. ∆H +endo/-exo
o. AB = k versus A/B = k
p. mole fractions and Dalton's Law
q. Boyle's, Charles', Gay Lussac, PV = nRT
r. kinetic energy (KE) = (1/2)mv2
s. Absolute zero, STP, and converting between Cº and K
t. ∆Hfus or ∆Hvap and the solid-melting-liquid-boiling-gas stair-step energy diagram
u. ∆BP = m KBP or ∆MP = m KMP
v. pH, pOH, [H+], [OH-], Kw
w. strong acids, strong bases
x. Kex = [products]/[reactants], but only (aq) and (g)
y. LEO says GER, and "An ox and red cat"
z. Eºcell = Eºred (reduced element) - Eºred (oxidized element)
Due:
Assignment
HW Collection & Review Sheet (due in basket, Thurs, May 22, before bell rings)
HW Collection (packet):
1. May 21: Balancing Redox D
2. May 23: Review - Ch. 17 Oxidation & Reduction
Review Sheet (salmon)
Finish RS. Unit 12 - Reduction & Oxidation
(Ch. 17)
Major Topics:
1. Predict oxidation #s for each element in a species
2. Use oxidation #s to determine which element is oxidized/reduced
3. Determine the oxidizing/reducing agents
4. Balance redox equations
5. Label the anode, cathode, and direction of e- flow in a voltaic cell
6. Describe the purpose of a salt bridge and direction of ion flow
7. Calculate Eº for a redox reaction (and is the reaction spontaneous?)
Labs:
1. Iron Transitions
-identifying redox
2. Metal Activity
-oxidized/reduced elements
-reducing/oxidizing agents
-redox trend in the metal activity series
Due:
Tues, 5/22/18 CW (stamped Wed, 5/23/18)
"Review - Ch. 17 Oxidation/Reduction" WS.
*Cell Notation Notes attached.
Check!
1a. H +1, Cl +5, O -2
1b. Sb +3, O -2
1c. As +5, O -2
1d. H +1, B +3, O -2
2a. C; I; I2O5; CO
2b. P; S; S8; P4
2c. I; Sb; SbCl5; KI
3a.* 8NO3 + 3Cu --> 3Cu(NO3)2 + 2NO + 4H2O
3b.* 2Bi(OH)3 + 3K2SnO2 --> 2Bi + 3K2SnO3 + 3H2O
4a. 14H+ + Cr2O72- + 3C2O42- --> 6CO2 + 2Cr3+ + 7H2O
4b. 3ClO2 + 4H+ + 4MnO4- --> 4MnO2 + 2H2O + 3ClO4-
basic: 3ClO2 + 2H2O + 4MnO4- --> 4MnO2 + 4OH- + 3ClO4-
5a. -Eºcell = not spontaneous (will not occur as written)
5b. +Eºcell = spontaneous (will occur as written)
6a. 3Fe2+ + 2Al --> 2Al3+ + 3Fe; Eº = 1.22 V
6b. Cl2 + Zn --> Zn2+ + 2Cl-; Eº = 2.12 V
7b. Ox: Cr --> Cr3+ + 3e-; Red: Cu2+ + 2e- --> Cu
7c/d/e. electrons flow from -anode (Cr) to +cathode (Cu)
7f. 1.08 V (spontaneous, voltaic)
7g. salt anions to anode beaker; salt cations to cathode beaker
8b. Ox: Pb --> Pb2+ + 2e-; Red: Zn2+ + 2e- --> Zn
8c/d/e. electrons flow from -anode (Pb) to +cathode (Zn)
8f. -0.63 V (not spontaneous, electrolytic)
8g. salt anions to anode beaker; salt cations to cathode beaker
*challenge (not what you'd see on an exam)
Due:
Mon, 5/21/18 CW (stamped Tues, 5/22/18)
"Balancing Redox C" WS, especially backside #5.
*Notes/sample problems attached.
Check! (pic attached)
Due:
Assignment
Fri, 5/18/18 CW (stamped Mon, 5/21/18)
"Balancing Redox D" WS.
Check!
1. 2Fe3+ + H2S --> 2Fe2+ + S + 2H+
2. ClO3- + 2MnO2 + 2OH- --> Cl- + 2MnO4- + H2O
3. 4H+ + Cu + SO42- --> Cu2+ + SO2 + 2H2O
4. 4OH- + SO2 + Br2 --> SO42- + 2H2O + 2Br-
5. 2H2O + 3P + 5NO3- --> 3PO43- + 4H+ + 5NO
6. 6Mn3+ + 6OH- + I- --> IO3- + 3H2O + 6Mn2+
7. 2Mn2+ + 14H+ + 5BiO3- --> 2MnO4- + 5Bi3+ + 7H2O
8. 10OH- + 2Cr3+ + 3ClO- --> 2CrO42- + 3Cl- + 5H2O
Please let me know if there are any typos above!
Due:
Thurs, 5/17/18 CW (stamped Fri, 5/18/18)
"Balancing Redox B" WS.
*Notes/samples attached.
Samples are "Balancing Redox A" #2 and "Balancing Redox B" #1.
Check:
1. 4H2O + 2NO3- + 6I- --> 2NO + 3I2 + 8OH-
2. 2H2O + C + 2SO42- --> CO2 + 2SO2 + 4OH-
3. H2O + 2NO3- + 2Sb --> Sb2O3 + 2NO + 2OH-
4. 8NH3 + 6NO2 --> 7N2 + 12H2O
Due:
Wed, 5/16/18 CW (stamped Thur, 5/17/18)
"Balancing Redox A" WS.
*Notes/samples attached.
Check!
1. 4H+ + 6NO3- + S --> 6NO2 + 2H2O + SO42-
2. 2H2O + 2Cr3+ + 3 MnO2 --> 2CrO42- + 3Mn2+ + 4H+
3. 6H+ + 2MnO4- + 5H2S --> 2Mn2+ + 5S + 8H2O
4. S + 2NO3- --> 2NO + SO42-
5. 5H+ + 5NO3- + P --> 5NO2 + H2O + H3PO4
6. 8H+ + Cr2O72- + 3H2S --> 2Cr3+ + 7H2O + 3S
7. 4H+ + MnO2 + 2Cl- --> Mn2+ + 2H2O + Cl2
8. 16H+ + 2MnO4- + 10Br- --> 5Br2 + 2Mn2+ + 8H2O
Due:
Assignment
Tues, 5/15/18 CW/HW (due in basket before bell rings, Wed, 5/16/18)
Lab - Single Replacement REDOX-style.
Due:
Mon, 5/14/18 CW (stamped Tues, 5/5/18)
"Identifying Redox" WS.
*Notes attached.
Due:
Assignment
Fri, 5/11/18 HW (submitted Mon, 5/14/18 before 7:59 AM)
1. Take notes by hand.
2. Compose your summary (on a Google doc to save your work if you want).
3. Copy-and-paste your summary here, choosing "Oxidation and Reduction."
Due:
Assignment
Wed-Thurs 5/9-5/10 (due Fri, 5/11/18)
Wednesday, May 9, 2018
Iron's Transitions gDoc here. Make one copy for your group and share with me ([email protected]).
Complete your procedure--approval pending.
Thursday, May 10, 2018
If your procedure is approved, commence with your experiment.
DUE Fri, 5/11/18
-turnitin.com by 7:59 AM
-one hard copy in basket for grading
Due:
Assignment
HW Collection & Review Sheet (due in basket, Tues, May 8, before bell rings)
HW Collection (packet):
1. Apr 24: Rate Laws using Initial Rates
2. Apr 30: Shifty Reaction
3. May 1: Kc expression
4. May 4: Ki/Ka/Kb
Review Sheet (lavender)
Finish RS. Unit 11 - Chemical Equilibrium
(Ch. 16)
Test on Tuesday, May 8.
Major Topics:
1. How reaction rate is affected by surface area, concentration/pressure, temperature, type of reactant, and the presence of a catalyst
2. Determine the rate law using concentration/rate data
3. Explain the process of equilibrium in terms of reaction rates
4. Write an equilibrium expression
5. Calculate K given equilibrium concentrations
6. Use Le Chatelier to predict the effect on equilibrium when concentrations, temperature, or pressure is changed
7. Calculate the pH of a weak acid solutions given initial concentration and Ka (and vice versa)
Labs:
1. Iodine Clock
-collision theory and factors of reaction rates
-rate laws using initial rates data
2. Le Chatelier
Due:
Assignment
Get your progress report signed! Due in basket, Tuesday, May 8, 2018.
Due:
Assignment
Fri, 5/4/18 HW (submitted Mon, 5/7/18 before 7:59 AM)
1. Take notes by hand.
2. Compose your summary (on a Google doc to save your work if you want).
3. Copy-and-paste your summary here, choosing "Batteries."
Due:
Wed, 5/3/18 CW (stamped Thur, 5/4/18)
Finish "Ki/Ka/Kb" WS.
*Notes attached.
Due:
Assignment
Wed, 5/2/18 HW (submitted Thurs, 5/3/18 before 7:59 AM)
1. Take notes by hand.
2. Compose your summary (on a Google doc to save your work if you want).
3. Copy-and-paste your summary here, choosing "Strong vs. Weak Acids."
Due:
Assignment
Mon-Tues, 4/30 and 5/1/18 CW/HW (due in basket before bell rings, Wed, 5/2/18)
Lab - Le Chatelier.
Due:
Tues, 5/1/18 CW (stamped Tues, 5/1/18)
"Kc expression" WS finished in class.
*Notes attached.
Due:
Assignment
Fri, 4/27/18 CW (stamped Mon, 4/30/18)
Finish "Shifty Reaction" WS.
Due:
Assignment
Wed, 4/25/18 HW (submitted Thurs, 4/26/18 before 7:59 AM)
1. Take notes by hand.
2. Compose your summary (on a Google doc to save your work if you want).
3. Copy-and-paste your summary here, choosing "Le Chatelier's Principle."
Due:
Mon, 4/23/18 CW (stamped Tues, 4/24/18)
Finish "Rate Laws using Initial Rates" WS.
*Remember to solve for all exponents and k.
*Notes/Samples attached.
Check!
3. 0.011 min-1 mM-2 [A][B]2
4. 1.1 x 10-5 M s-1
5. 4.19 x 10-3 M s-1
6. 0.1 M-2 min-1 [A]2[B] if rates are M min-1
7. 6.7 M-1 s-1 [CH3CHO]2
8. 13 M-3 min-1 [Q][X]3
9. 7.0 x 103 M-2 s-1 [NO]2[O2]
10. k[A][B]2
11. effect on rate is x20.5
Due:
Assignment
Thurs-Fri 4/19//18-4/20/18 Lab (due Mon, 4/23/18)
Thursday:
1. Each group makes ONE copy of this Google Doc: "Lab - Iodine Clock."
-Share with me.
2. Observe and time demos (Trials 1, 2, 3)
3. Create graphs.
Friday:
1. Implement your 4th and 5th trials w/ approved volumes for your last two data points.
2. Finalize your graphs.
3. Get your challenge run timed.
Before the Monday deadline:
a. One group member should submit to turnitin.com (before 7:59 AM) when finished.
b. One group member should print a hard copy for the basket (before the bell rings) on Monday.
Due:
Assignment
HW Collection & Review Sheet (due in basket, Wed, Apr. 18, before bell rings)
HW Collection (packet):
1. Apr 11: Titrations
2. Apr 12: POGIL - Calculating pH (p. 3-4)
3. Apr 13: pH (+separate sheet of paper)
4. Apr 17: Ionic Equations
Review Sheet (lime green)
Finish RS. Unit 10 - Acids and Bases
(Ch. 13)
Test on Wednesday, April 18.
Major Topics:
1. Determine if a solution is acidic or basic using (a) chemical properties; (b) pH; (c) common indicators (litmus)
2. Determine the concentration of a solution or molar mass of a solute using titration info (i.e. stoichiometry)
3. Explain an acid-base reaction in terms of the Arrhenius Theory and the Bronsted-Lowry Theory
4. Calculate the pH, POH, [H+], and [OH-] given one of the four
5. Difference between strong and weak electrolytes
6. Convert a molecular equation to a net ionic equation
Labs:
1. Acid Base Characteristics
-double-replacement reactions (inc. neutralization)
-acid and base litmus colors
2. Vinegar Titration
-solve for an unknown concentration using stoich
3. Cabbage Lab
-change in pH vs. change in concentration H+ or OH-
Due:
Mon, 4/16/18 CW (stamped Tues, 4/17/18)
Finish "Ionic Equations" WS.
Memorize your strong acids: HCl, HBr, HI, HNO3, H2SO4, HClO3, HClO4
*Notes/Samples attached.
CHECK:
3. Sr2+ + SO42- --> SrSO4(s)
4. Mn2+ + CO32- --> MnCO3(s)
5. NR
6. Al3+ + PO43- --> AlPO4(s)
7. Cu+ + I- --> CuI(s)
8. NR
9. 2Co3+ + 3S2- --> Co2S3(s)
10. 3Ba2+ + 2PO43- --> Ba3(PO4)2(s)
11. Fe3+ + 3OH- --> Fe(OH)3(s)
12. NR
Due:
Assignment
Fri, 4/13/18 CW/HW (due in basket before bell rings, Mon, 4/16/18)
Lab - pH and Cabbage Juice.
Due:
Assignment
Wed, 4/11/18 CW/HW (due in basket before bell rings, Fri, 4/13/18)
Lab - Titration.
Due:
Thurs, 4/12/18 CW (pH WS stamped Fri, 4/13/18)
1. Finish POGIL "Calculating pH" in class.
2. pH, pOH, [H3O+], [OH-] and SigFig notes
3. Finish "pH" WS.
*Notes/Sample #2,3 attached.
CHECK:
work must be shown (always)
4.49 pH, 9.51 pOH, 3.1E-10 M OH-, acid
11.17 pH, 2.83 pOH, 1.5E-3 M OH-, base
1.9E-6 M H+, 8.27 pOH, 5.4E-9 M OH-, acid
1.4E-9 M H+, 5.14 pOH, 7.2E-6 M OH-, base
2.4E-8 M H+, 7.62 pH, 4.2E-7 M OH-, base
3.3E-2 M H+, 1.48 pH, 3.0E-13 M OH-, acid
2.1E-11 M H+, 10.68 pH, 3.32 pOH, base
5.3E-7 M H+, 6.28 pH, 7.72 pOH, acid
1. 3.1E-11 M OH-; 3.49 pH; 10.51 pOH; acid
2. 1.6E-12 M H+; 11.79 pH; 2.21 pOH; base
3. 4.8E-12 M H+; 2.1E-3 M OH-; 2.68 pOH; base
4. 1.9E-5 M H+; 5.4E-10 M OH-; 4.73 pH; acid
Due:
Tues, 4/10/18 CW (stamped Wed, 4/11/18)
Finish "Titrations" WS.
*Notes/Samples attached.
CHECK:
1. 0.15 M KOH
2. 0.750 M NaOH
3. 0.29 M Ca(OH)2
4. 14.3 mL Al(OH)3
5. 29 mL H2CO3
6. 0.315 M HNO3
7. 1.104 M HCl
8. 176 g/mol HAsc
9. 150. g/mol H2Tart
Due:
Assignment
Fri-Mon, 4/6 and 4/9/18 CW/HW (due in basket before bell rings, Tues, 4/10/18)
Lab - Acids and Bases Properties.
Ingredients
a. Rolaids
-Active ingredients: calcium carbonate, magnesium hydroxide
-Inactive ingredients: dextrose, flavor, magnesium stearate, silicon dioxide, stearic acid, sucralose, sucrose
b. Sprite
-carbonated water, high fructose corn syrup, citric acid, natural flavors, sodium citrate, sodium benzoate
c. Coca-Cola
-carbonated water, high fructose corn syrup, caramel color, phosphoric acid, natural flavors, caffeine
Due:
Mon, 4/9/18 CW
*POGIL Acids and Bases Review/Notes attached
Due:
Assignment
HW Collection & Review Sheet (due in basket, Thurs, Apr. 5, before bell rings)
HW Collection (packet):
1. Mar. 6: % Solution
2. Mar. 7: p. 3-4 of Solubility POGIL
3. Mar. 12: Molarity
4. Mar. 13: Salt/Sugar Simulation
5. Mar 14/15: Dilution
6. Mar. 20: Solution Stoich
7. Mar. 21: Colligative Properties, Molality
8. Mar. 22: ∆TBP and ∆TFP
9. Mar. 23: Colloids, Suspensions
Review Sheet (ivory)
Finish RS. Unit 9 - Solutions
(Ch. 13)
Test on Thursday, April 5.
Review - Ch. 13 (opt) KEY
11. supersaturated; unsaturated; supersaturated
---
1. 0.397 m CaCl2 in CH3OH
2. 4.04% MgCl2
3. 0.719 g NaCl
4. 0.0802 M BaI2
5. 24 g LiNO3
6. -1.63ºC
7. 200 g/mol
8. Start w/ 13 mL of 3.0 M CuSO4, then add water (237 mL) until the new volume 250. mL is reached.
9. 0.311 M AgNO3
10. 801 mL HCl soln
Major Topics:
1. Predict whether two liquids are miscible/immiscible based on polarity
2. Determine if a solution is unsaturated, saturated, or supersaturated
3. Calculate mass percent, molarity, and molality of a solution
4. Calculate the freezing/boiling point of solution and molar mass of a substance using colligative property data
5. Understand how gas solubility is affected by pressure and temperature
6. Understand how solid solubility is affected by temperature, surface area, and stirring
7. Understand colloids vs. solutions/suspensions using properties like the Tyndall Effect
Due:
Assignment
Wed, 4/4/18 HW (submitted Thurs, 4/5/18 before 7:59 AM)
1. Take notes by hand.
2. Compose your summary (on a Google doc to save your work if you want).
3. Copy-and-paste your summary here, choosing "Acids and Bases."
Due:
Assignment
Mon-Tues, 4/2 and 4/3/18 CW/HW (due in basket before bell rings, Wed, 4/4/18)
Lab - Cranberry.
Due:
Fri, 3/23/18 CW
*Notes attached + video of a supersaturated solution
Due:
Wed, 3/21/18 CW (stamped Thurs, 3/22/18)
Finish "∆Tf and ∆Tb" WS.
*Notes/Samples attached.
**WS 2.0 posted.
***Work shown from classwork.
CHECK:
1. -4.35ºC and -4.4ºC
2. 0.27 m I2 in C6H6
3. 0.87ºC
4***. 113 g/mol
5. 65 g/mol
6. CH2O; 180 g/mol; C6H12O6
Due:
Tues, 3/20/18 CW (stamped Wed, 3/21/18)
Finish "Colligative Properties and Molality" WS.
*Notes/Sample/Key attached.
Due:
Assignment
Mon, 3/19/18 CW (stamped Tues, 3/20/18)
Finish "Molarity Stoichiometry" WS.
Due:
Assignment
3/15/18 CW/HW
1. Make a copy of this Google doc for your group.
-Make sure everyone contributes; I expect to see everyone's color represented evenly
2. Share document with me ([email protected])
3. Plan out Pre-Lab with your group (everyone grab Chromebooks)
Notes:
Recall that solubility is the maximum amount of solute that can be dissolved in a certain amount of solvent at a specific temperature. This is different from Rate of Solubility. Three factors that affect the rate of solubility are (1) temperature, (2) agitation (e.g. stirring), and (3) surface area (e.g. grain size, which is inversely related to surface area). Note: The word "rate" always includes a measurement of time; less time means faster process, and more time means slower process.
3/16/18 Check-in
1. Get your Procedure approved.
-(or fix your Procedure until approved)
With your group:
2. Implement your Procedure
3. Complete lab writeup
4. Designate someone to Submit to turnitin.com before Mon. 7:59 AM
5. Designate someone to Print one (1) hard copy per group for basket
Lab is due in basket, Mon, 3/19/18 before the bell rings.
Due:
Per 1, 3, 6 Tues, 3/13/18 CW (stamped Wed, 3/14/18)
Per 2 Wed, 3/14/18 CW (stamped Thur, 3/15/18)
Finish "Dilution" WS.
*Sample problem attached.
CHECK!
1. 0.14 M NaCl
2. 0.570 g NaOH
3. 0.18 mol HCl; 100.0 mL ?M HCl soln; No, there are still 0.18 mol HCl present in the 100.0 mL soln
4. 25 mL 2.0 M CuSO4
5. 0.96 M
6. 20 mL H2O
7. Start w/ 130 mL 3.0 M AgNO3, then add 270 mL H2O to reach 400. mL
8. 0.125 L 3.00 M KI
9. 0.19 M NaBr
10. 0.033 M LiC2H3O2
11. 0.63 M NaCl; add 900 mL H2O to the 600 mL 0.63 M NaCl to make 1500 mL 0.25 M NaCl
Due:
Assignment
Mon, 3/12/18 CW (stamped Tues, 3/13/18)
Finish "Salt and Sugar Simulations." Link is at the head of the packet.
Due:
Assignment
Fri, 3/9/18 CW (stamped Mon, 3/12/18)
Finish "Molarity" WS.
CHECK!
10. 6.85 g Ni(NO3)2
11. 547 mL (NH4Cl) soln
12. 556 mL (K2CrO4) soln
13. 137 mL (KNO3) soln
14. 0.0789 M Ca(OH)2
Due:
Assignment
HW Collection & Review Sheet (due in basket, Thurs, Mar. 8, before bell rings)
HW Collection (packet):
1. Feb. 14: IMF HW (+separate sheet of paper)
2. Feb 22: Heating Curve Practice (+separate sheet of paper)
3. Feb 23: Hydrates
4. Mar. 1: Vapor Pressure and Boiling Point (CW)
5. Mar. 2: Viscosity and Surface Tension
Review Sheet (lime green)
Finish RS. Unit 8 - Solids and Liquids
(Ch. 11)
Test on Thursday, March 8.
Major Topics:
1. Describe the gas, liquid, and solid state
2. Describe the 4 types of crystalline solids (ionic, molecular, network covalent, metallic)
3. Distinguish between different IMFs (i.e. for molecular solids)
4. Interpret MP and BP graphs in terms of IMFs
5. Relate properties (heat of fusion/vaporization, vapor pressure, MP/BP, viscosity, surface tension) of liquids to relative strengths of IMFs
6. Calculate the energy associated with phase changes
7. Describe a water molecule and water's physical and chemical properties
8. Name hydrates and calculate water content from hydrate formula (and vice versa)
2. Describe the 4 types of crystalline solids (ionic, molecular, network covalent, metallic)
3. Distinguish between different IMFs (i.e. for molecular solids)
4. Interpret MP and BP graphs in terms of IMFs
5. Relate properties (heat of fusion/vaporization, vapor pressure, MP/BP, viscosity, surface tension) of liquids to relative strengths of IMFs
6. Calculate the energy associated with phase changes
7. Describe a water molecule and water's physical and chemical properties
8. Name hydrates and calculate water content from hydrate formula (and vice versa)
Review - Ch. 13 Liquids, Solids, and Water
1b. @25ºC: liquid methanol, gas oxygen, liquid water, solid silver
2. 150 cal + 2.0 x 103 cal + 2.50 x 103 cal + 13500 cal = 18200 cal
3. Physical, because chemical bonds are not broken or formed. More energy is needed to completely separate the molecules in liquid to gas state, as opposed to less energy to simply reduce the IMFs in solid to liquid state.
4. by column:
atoms, ions, molecules/atoms
high, high, low
high, high low
malleable, brittle, brittle
iron, brass; salt, rust; iodine, ice
5. by column:
2, 3, 1(depends, very conditional: if... then...)
inverse
direct
direct
direct
6. dipole-dipole, dispersion
dipole-dipole, dispersion
dipole-dipole, H-Bond, dispersion
dispersion
dipole-dipole, dispersion
7. barium hydroxide octahydrate; SnCl4•5H2O; FePO4•4H2O; calcium sulfate monohydrate
8. 47.25% H2O; cobalt (II) sulfate monohydrate
9. by column:
def, var, var
def, def, var
def, def, var
no, no, yes
no, no, yes
Due:
Assignment
Tues, 3/6/18 CW (stamped Wed, 3/7/18)
Finish p.3-4 of POGIL - Solubility.
Due:
Assignment
Fri-Mon, 3/2 and 3/5/18 CW/HW (due in basket before bell rings, Tues, 3/6/18)
Lab - Evaporation and IMFs.
Due:
Mon, 3/5/18 CW (stamped Tues, 3/6/18)
Finish "% Solution Problems" WS.
*Notes and #1-2 attached.
CHECK!
1. 2.78% ethanol
2. 7.9 g lactose
3. 5.0% HC2H3O2
4. 53.6 g solution
5. 2.50 g HNO3
6. 1.3 g KCl
7. 15.8 g alcohol
8. 67.0 g HCl solution
Due:
Thurs, 3/1/18 CW (stamped Fri, 3/2/18)
Finish "Viscosity and Surface Tension" WS.
*Notes for #4-5 of "Pvap & BP" CW attached.
Due:
Tues, 2/27, Thurs 3/1/18 CW (stamped Thurs, 3/1/18)
"Vapor Pressure and Boiling Point" WS finished in class.
*Notes "Evaporation" and warmup (#1-3) attached.
Due:
Assignment
Mon-Wed, 2/26-2/28/18 CW/HW (due in basket before bell rings, Thurs, 3/1/18)
Lab - Heat of Fusion.
Due:
Assignment
Fri, 2/23/18 (due in basket Mon, 2/26/18)
Sign progress report.
Due:
Fri, 2/23/18
Progress report due in basket, Monday, 2/26/18.
*CER "Evidence+R" warmup attached.
Due:
Thurs, 2/22/18 CW (stamped Fri, 2/23/18)
Finish "Hydrates" WS.
*Notes attached.
Select answers:
3. 40.55% water, 14.40% water
4. nickel (II) chloride dihydrate; chromium (III) iodide nonahydrate; calcium nitrate tetrahydrate
Due:
Wed, 2/21/18 CW (stamped Thurs, 2/22/18)
Finish "Heating Curve problems" WS.
*Notes attached.
*CER "Evidence" warmup attached.
Due:
Assignment
Tues, 2/20/18 HW (submitted Wed, 2/21/18 before 7:59 AM)
1. Take notes by hand.
2. Compose your summary (on a Google doc to save your work if you want).
3. Copy-and-paste your summary here, choosing "BPs and IMFs."
Due:
Tues, 2/13/18 CW (stamped Wed, 2/14/18)
Finish "IMF HW" WS.
*Notes attached.
Remember: ionic, covalent, and metallic bonds are INTRAmolecular forces, not IMFs.
Types of Solids POGIL notes:
4 Types of Solids
a. Ionic (ionic bonds hold ions in solid state)
b. Molecular (IMFs hold molecules in solid state)
c. Network Covalent (covalent bonds hold atoms/molecules in solid state)
d. Metallic (metallic bonds hold atoms in solid state)
7. Strength of attractive force is directly related to the energy required to break said force, which explains many properties like MP/∆Hfus and BP/∆Hvap
12-14. Solubility depends on the rule "like dissolves like," such as (a) polar substances dissolving ionic/polar substances or (b) nonpolar substances dissolving nonpolar substances.
Conductivity requires the substance to have "charged particles in a mobile state."
Due:
Assignment
Mon, 2/12/18 HW (submitted Tues, 2/13/18 before 7:59 AM)
1. Take notes by hand.
2. Compose your summary (on a Google doc to save your work if you want).
3. Copy-and-paste your summary here, choosing "Types of Solids."
Due:
Assignment
HW Collection & Review Sheet (due in basket, Fri, 2/9/18)
HW Collection (packet):
1. Feb 2: Boyle, Charles, Gay-Lussac, Combined Gas Laws
2. Feb 2: Gas Laws and Stoichiometry
3. Feb. 5: Gas Density and Molar Mass
4. Feb. 6: Practice... (Dalton's Law of Partial Pressures and Mole Fractions)
Review Sheet (orange)
Finish RS. Unit 7 - Gases
(Ch. 10)
Test on Friday, 2/9/18
Major Topics:
1. Properties of Gas
2. Convert between common units of P (atm, torr, mmHg)
3. Use Boyle's, Charles', Gay-Lussac's, or Combined gas law to calculate a new P, V, or T after a change in conditions.
4. Explain gas variables changes in terms of the kinetic molecular theory.
5. Use Ideal Gas Law to find P, V, n, or T given any three of the four variables.
6. Use Ideal Gas Law to convert between molar mass and density.
7. Use Dalton's law to calculate the total pressure of a mixture of gases.
8. Use Dalton's law to correct for water vapor pressure when collecting a gas over water.
Due:
Assignment
Tues-Wed, 2/6 and 2/7/18 CW/HW (due in basket before bell rings, Thurs, 2/8/18)
Lab - Target Gas.
Due:
Mon, 2/5/18 CW (stamped Tues, 2/6/18)
Finish "Practice..." WS* on Dalton's Law of Partial Pressures and Mole Fractions.
*Notes attached.
Due:
Fri, 2/2/18 CW (stamped Fri, 2/2/18)
Finish "Gas Laws and Stoichiometry" WS*.
Fri, 2/2/18 CW/HW (stamped Mon, 2/5/18)
Finish "Gas Density and Molar Mass" WS*.
*Notes and Keys attached.
Due:
Assignment
Thurs, 2/1/18 CW/HW (stamped Fri, 2/2/18)
Finish "Boyle, Charles, Gay-Lussac, Combined Gas Laws" WS.
TYPO:
9. 301 mL
Due:
Wed, 1/31/18 CW/HW (due Thurs, 2/1/18)
Finish notes on gas laws: "Boyle, Charles, Gay-Lussac, Combined."
Reminder:
STP = Standard Temperature and Pressure
a. (5sf) Standard Temperature = 0 ºC = 273.15 K
b. (∞sf) Standard Pressure = 1 atm = 760 mmHg = 760 torr
Due:
Assignment
Tues-Wed, 1/30 and 1/31/18 CW/HW (due in basket after class, Wed, 1/31/18)
Lab - Gay-Lussac's Law.
Due:
Fri-Mon, 1/26 and 1/27/18 CW/HW (due in basket before bell rings, Tues, 1/30/18)
Lab - Boyle's Law.
Notes
1. Pressure Conversions
2. Kinetic Molecular Theory (KMT)
Due:
Assignment
Thurs, 1/25/18 HW (submitted Fri, 1/26/18 before 7:59 AM)
1. Finish the POGIL on "Gas Variables."
2. Submit your responses HERE for homework credit. The collectively surveyed data will be used to assess which concepts need re-teaching on Monday.
Due:
Assignment
HW Collection & Review Sheet (due in basket, Wed, 1/24/18)
HW Collection (packet):
1. Dec 19: Periodic Trends and Polarity
2. Jan 10: Dot structures/structural formulas
3. Jan 16: Molecular Geometry 1-5
4. Jan 17: Molecular Geometry (mixed)
5. Jan 18: Heat of Formation
6. Jan 19: Enthalpy Stoich
7. Jan 23: Lab - Endo/Exo Reactions
Review Sheet (yellow)
Finish RS. Unit 6 - Periodic Trends, VSEPR, & Enthalpy
(Ch. 5, 12, and 9)
Test on Wednesday, 1/24/18
1. Periodic Table (Ch. 5)
a. How to predict the trend in atomic/ion size, ionization energy, and electronegativity for groups and periods
2. Chemical Bonding (Ch. 12)
a. Explain the difference between ionic bonds, covalent bonds, formula units, molecules
b. Draw electron dot structures for molecules and polyatomic ions
c. Apply VSEPR theory and predict the shapes of molecules and the bond angles
d. Identify the polarity of a bond using delta notation
e. Predict whether a bond is nonpolar, polar, or ionic using electronegativity
f. Determine if a molecular is polar by analyzing the symmetry of the geometry
3. Chemical Equation Calculations (Ch. 9)
a. Identify an endothermic/exothermic reaction using ∆H
b. Calculate the ∆H for a reaction using heats of formation
c. Solve mass/heat problems
Due:
Assignment
Fri, 1/19/18 CW/HW (due in basket before the bell rings, Mon, 1/22/18)
Get started on the calculations on the back of the Endo/Exo Lab. See Dec. 5 homework ("Stoichiometry") for guidance.
Due:
Assignment
Thurs, 1/18/18 CW/HW (stamped Fri, 1/19/18)
Finish
a. Heat of formation
b. Enthalpy Stoichiometry
WS.
CHECK:
| 1. | 2. | 3. | 4. | 5. | |
| ∆Hfº | -133.8 kJ | -566.0 kJ | -890.3 kJ | -1123.6 kJ | -113 kJ |
| ∆H Stoich | -79.5 kJ | -155 kJ | 17.4 kJ | -3.70 kJ | -141 kJ |
Due:
Assignment
Tues, 1/16/18 CW/HW (stamped Wed, 1/17/18)
Finish "Molecular Geometry (mixed)" WS.
Due:
Wed, 1/17/18 CW
Finish your notes on "Endothermic vs. Exothermic" and "Enthalpy."
Due:
Assignment
Fri, 1/12/18 CW/HW (stamped Tues, 1/16/18)
Finish "Shapes 1-5" WS.
Due:
Assignment
Wed, 1/10/18 CW/HW (stamped Thurs, 1/11/18)
Finish* "VSEPR Theory" WS.
*List three things you have learned from this activity.
Due:
Tues, 1/9/18 CW/HW (stamped Wed, 1/10/18)
2. "How to Draw Dot Structures" Notes
3. Use your notes to practice dot structures.
HW: Finish "Dot structures/structural formulas" WS.
Check your notes (pic attached)
Due:
Assignment
For SEM1 FINAL EXAM
Once upon a time, you memorized many things: a. prefixes (kilo, deci, centi, milli, micro, nano) b. D = m/V c. q = mC(delta T) d. Elements/Symbols (pink sheet) e. Conservation of mass and energy f. 1s22s22p63s23p6... (think apartment complex sheet) g. ROYGBIV and the EM spectrum h. Atomic/ionic radius, ionization energy, and electronegativity trends i. Charges for monoatomic and polyatomic ions j. Chemical equation subtypes k. Avogadro's # and 22.4 L l. % yield = actual/theoretical masses x 100% m. Rules for each category of significant figures and naming/formulas n. VSEPR o. Electronegativity ranges for bond types p. Celsius <--> Kelvin conversion (+/- 273.15) The final review practice is not collected. a. For question 7, use 2.70 g/cm3 for aluminum's density. b. For question 8, use 0.92 g/mL for ice's density. c. For question 10, use 0.0923 cal/(g x degree Celsius) for copper's specific heat capacity. d. For question 16, use 1.00 cal/(g x degree Celsius) for water's specific heat capacity. KEY1. 1.4E7 nm, 1600 g, 0.15 cubic meters, 1.9E4 mL, 1.5 L, 926 mi/hr2. 4, 1, 4, 23. 147, 0.21, 5.7E105. 1 m = 10 dm; 1 m = 100 cm; 1 m = 1000 mm; 1 m = 1E6 micrometers; 1 m = 1E9 nm; 1 m = 1E-3 km or 1000 m = 1 km6. 19 g/mL7. 17000 cubic cm Al8. 5800 g10. 1.0E3 cal15. 0.160 g oxygen16. 0.17 cal/(g x degree C)22. 92 protons, 146 neutrons, 92 electrons23. 12 protons, 14 neutrons, 10 electrons25. 28.11 amu26. 4, 10, 2, 3228. 1s22s22p63s23p2; 1s22s22p63s23p64s23d104p65s229. [Kr-36]5s24d105p5; [Xe-54]6s24f145d930. 4, 232. +2, -333. Pd34. Cr & Fe35. iodine36. Mg37. As, Pt38. P, Br39. F, Al40. Se, C41. SO2, CO2, SiBr442. hydrochloric acid; chlorous acid; perchloric acid; potassium sulfide; iron (III) oxide; copper (II) iodide; nickel (III) sulfate; silver nitrate; sodium acetate; sulfur trioxide43. H2S; H2SO4; H2SO3; Mg3N2; Au(NO2)3; CoBr3; Zn3(PO4)2; XeF444. 63.55 g/mol Cu; 199.88 g/mol CaBr245. 0.157 mol Cu; 2.2E23 molec CO2; 3.6E22 f.un Ag2S; 2.0 L O246. 35.00% N47. 3.17 g/L48. Cl2O749. C4H5O250. 3Mg + N2 --> Mg3N2; 2HgO --> 2Hg + O2; H2SO4 + 2KOH --> 2H2O + K2SO4; Mg + NiCl2 --> MgCl2 + Ni; Cl2 + 2NaI --> 2NaCl + I2; 2KClO3 --> 2KCl + 3O2; 2AgNO3 + Na2CO3 --> Ag2CO3(s) + 2NaNO351. 0.750 mol HCl; 0.230 g H2; 137 g AlCl352. 98.8%53. Fe limits, 5.72 g Fe2O3 form, 0.28 g O2 remain54. -58.9 kJ, exothermic55. 618 kJ58. 259. trigonal pyramid (107 degrees); bent (120 degrees); square planar (90 degrees)60. NaI is ionic; P-Br (partial + for P, partial - for Br); C-F (partial + for C, partial - for F); Cl-Br is nonpolar61. PCl3, SO2
Due:
Assignment
Mon, 12/18/17 CW/HW (stamped Tues, 12/19/17)
Finish "Periodic Trends and Polarity" WS.
Due:
Assignment
Fri, 12/15/17 CW/HW (stamped Mon, 12/18/17)
Finish "Review - Ch. 5" WS.
Due:
Assignment
12/8/17 HW (due in basket, Tues, 12/12/17)
Finish light pink RS. Unit 5 - Chemical Reactions & Stoichiometry.
Test on Tuesday, 12/12/17
1. Chemical Reactions (Law of Conservation of Mass)
a. recognize and balance chemical equations
b. predict the products of a combination, decomposition, single replacement, double displacement (precipitation, neutralization), and combustion reaction by writing a correctly balanced chemical equation
c. arrange elements in order of increasing activity
d. identify precipitates in double displacement reactions
2. Stoichiometry (Law of Definite Proportions, Conservation of Matter & Energy)
a. solve variety of problems including mass/mass, mass/volume, mole/mole, volume/volume, and limiting reactant
b. calculate the percent yield given the actual yield and the theoretical yield
Practice Problems
Chemical Reactions
1. C; 1, 1, 1
2. CB; 1, 8, 5, 6
3. DR; 1, 2, 2, 1
4. D; 2, 1, 1, 1
5. SR; 1, 2, 1, 2
6. DR; 1, 1, 1, 1
7. CB; 1, 2, 1, 2
8. SR; 1, 1, 1
9. C; 1, 1, 1
TYPO! SO2 + H2O --> H2SO3
10. D; 2, 2, 1
11. combination --> NaCl; 2, 1, 2
12. single replacement --> NaOH + H2; 2, 2, 2, 1
13. combustion --> CO2 + H2O; 1, 3, 2, 3
14. decomposition --> KCl + O2; 2, 2, 3
15. double replacement --> HOH + MgCl2; 2, 1, 2, 1
16. double replacement --> NH4Cl + CaCO3(s); 1, 1, 2, 1
17. combination --> AlBr3; 2, 3, 2
18. decomposition --> H2 + O2; 2, 2, 1
19. single replacement --> NaCl + I2; 1, 2, 2, 1
20. combustion --> CO2 + H2O; 1, 5, 3, 4
21. double replacement --> Ag2CrO4(s) + KNO3; 2, 1, 1, 2
22. decomposition --> CaO + CO2; 1, 1, 1
23. combustion --> CO2 + H2O; 2, 15, 12, 6
24. single replacement --> AlCl3 + H2; 2, 6, 2, 3
25. combination --> Na2SO4; 1, 1, 1
Stoichiometry
1. 5.73 L H2
2. 102 g ZnCl2
3. 36.4 g PbCl2
4. 22.9 g NaCl with 15.5 g Cl2 left over
5. 96.7% NO yield
Due:
Assignment
Mon, 12/11/17 CW/HW (due in basket before the bell rings, Tues, 12/12/17)
Final check of Lab - Double Replacement (Part 3) + Purpose.
Due:
Assignment
Wed, 12/6/17 CW/HW (due in basket before the bell rings, Fri, 12/8/17)
Finish Lab - Target Stoichiometry.
Due:
Assignment
Thurs, 12/7/17 CW/HW (stamped Fri, 12/8/17)
Finish "Limiting Reactant" WS.
CHECK:
1. NaOH (0.0628 mol NaOH < 0.0656 mol H2SO4)
2. HCl (0.0952 mol HCl < 0.101 mol Mg(OH)2)
3. 12.1 g CrCl3 produced with 0.14 g Cr left over
4. 17.0 g Fe
5. I2O5 (0.007910 mol I2O5 < 0.008674 mol SF4)
6. 23.4 g H2O produced with 11.7 g N2O4 left over
Due:
Assignment
Thur, 12/7/17 CW/HW (due in basket before the bell rings, Fri, 12/8/17)
2nd check of Lab - Double Replacement (Part 3).
Due:
Assignment
Tues, 12/5/17 CW/HW (stamped Wed, 12/6/17)
Finish #2-3 on "% Yield" WS.
CHECK:
1. 49.1% NaCl yield
2. 73.7% Sb yield
3. 78.3% NO yield
Due:
Assignment
Tues, 12/5/17 CW/HW (due in basket before the bell rings, Wed, 12/6/17)
Finish Lab - Double Replacement (Part 3).
Due:
Mon, 12/4/17 CW/HW (stamped Tues, 12/5/17)
Finish "Stoichiometry" WS. Balance on a separate piece of paper as needed.
CHECK:
1. 3.75 mol O2
2. 1.88 mol H2
3. 272 mL HI
4. 50.0 L SO2
5. 3.54 g S8
6. 1.55 g C6H6
7. 106 g AlCl3
8. 90.2 L H2
9. 0.386 g Sb
10. 10.1 g Al
Due:
Assignment
Fri, 12/1/17 HW (completed by Mon, 12/4/17)
Finish Lab - Double Displacement (Part 1 - The Predictions)
Due:
Thurs, 11/30/17 CW/HW (stamped Fri, 12/1/17)
1. Check your Single/Double Replacement notes (pic attached).
2. Finish "Single/Double Replacement" WS. Balance on a separate piece of paper as needed.
CHECK:
SR #1-11 pic attached.
DR #1-11 pic attached.
Due:
Assignment
Tues-Wed, 11/28and29/17 CW/HW (due in basket before the bell rings, Thurs, 11/30/17)
Finish Lab - Metal Reactivity.
Staple together:
1. Procedure
2. Data Table
3. Balanced equations
4. Analysis (e.g. questions, CER)
Due:
Mon, 11/27/17 CW/HW (stamped Tues, 11/28/17)
1. Finish the How to write products for your "5 Types of Reactions" notes.
2. Finish "Predicting Products" WS. Balance on a separate piece of paper as needed.
CHECK:
(SR) 1. MgBr2 + Cl2 --> MgCl2 + Br2
(SR) 2. 2Al + Fe2O3 --> 2Fe + Al2O3
(DR) 3. 2AgNO3 + ZnCl2 --> 2AgCl + Zn(NO3)2
(D) 4. CoCO3 --> CoO + CO2
(SR) 5. Zn + 2HCl --> H2 + ZnCl2
(DR) 6. H2SO4 + 2NaOH --> 2HOH + Na2SO4
(C) 7. 4Al + 3O2 --> 2Al2O3
(SR) 8. copper(II) needs to be given to you
2HC2H3O2 + Cu --> H2 + Cu(C2H3O2)2
(D) 9. 2KClO3 --> 2KCl + 3O2
(C) 10. CaO + HOH --> Ca(OH)2
(CB) 11. C9H20 + 14O2 --> 9CO2 + 10H2O
(DR) 12. Zn(NO3)2 + 2KI --> 2KNO3 + ZnI2
(DR) 13. --> 2NH4Cl + ZnS
(SR) 14. lead(II) needs to be given to you
--> Zn + PbCl2
(C) 15. --> MgO with 2,1,2 for balancing
(D) 16. --> Hg + O2 with 2,2,1 for balancing
(CB) 17. --> CO2 + H2O with 2,7,4,6 for balancing
(DR) 18. --> HOH + BaSO4 with 1,1,2,1 for balancing
(D) 19. --> MgCl2 + O2 with 1,1,3 for balancing
(SR) 20. --> Ca(OH)2 + H2 with 1,2,1,1 for balancing
Due:
Tues, 11/21/17 CW/HW (stamped Mon, 11/27/17)
1. Finish the Types of Reactants for your "5 Types of Reactions" notes.
2. Finish "Types of Chemical Reactions" WS. Balance on a separate piece of paper.
Due:
Assignment
11/9/17 HW (due in basket, Mon, 11/20/17)
Finish light blue RS. Unit 4 - Bonding, Nomenclature, & Moles.
Test on Monday, 11/20/17
1. Bonding
-explain the difference between ionic bonds and covalent bonds
-explain the difference between formula units and molecules
-describe the formation of an ionic bond between a metal atom and nonmetal atom
-distinguish between cations and anions
-describe the formation of a covalent bond between two nonmetal atoms
2. Naming/Formula Writing
-classify a compound as ionic, covalent, or acid
-classify a compound as binary or ternary
-identify the metal of an ionic compound as being a fixed charged metal or a variably charged metal
-correctly name covalent compounds, ionic compounds, and acids
-correct write the formula for covalent compounds, ionic compounds, and acids
3. Moles
-Avogadro's # (atoms, ions, molecules, formula units), molar mass
-convert between moles, particles, Liters of a gas at STP, and grams
-calculate density of a gas at STP using molar mass (and vice versa)
-calculate the % composition of a compound given the formula of the compound
-determine the empirical and molecular formula of the compound given the % composition
Due:
Assignment
Mon, 11/13/17 HW (due in basket before the bell rings, Mon, 11/20/17)
Finish Lab - Hydrates.
Due:
Assignment
Tues, 11/14/17 (optional)
Ch. 8 Review WS.
CHECK:
1. 109.00 g/mol CrF3, a.k.a. chromium (III) fluoride; 423.2 g/mol PbCr2O7, a.k.a. lead (II) dichromate; 132.17 g/mol (NH4)2SO4, a.k.a. ammonium sulfate
2. 78.58% Ni, 21.42% O, nickel (II) oxide; 31.91% K, 28.93% Cl, 39.17% O, potassium chlorate; 28.48% Ca, 22.01% P, 49.28% O, 0.239% H
3. 0.275 mol CO2, a.k.a. carbon dioxide; 6.16 L CO2
4. 5.75 x 1023 molec CH3OH
5. 154 L C2H6, a.k.a. dicarbon hexahydride; 207 g C2H6
6. BaCl2, a.k.a. barium chloride
7. Na2SiO3
8. P2O3, a.k.a. diphosphorus trioxide
9. C10H14N2
10. 2.49 g/L C4H10, a.k.a. tetracarbon decahydride; 4.65 g/L SiF4, a.k.a. silicon tetrafluoride; 1.78 g/L Ar
11. 16.0 g/mol methane; 65.6 g/mol SO2
Due:
Assignment
Thurs, 11/16/17 (stamped Fri, 11/16/17)
Finish "Balancing Equations" WS.
Due:
Fri, 11/17/17
Quadrant 3 of "5 Types of Reactions" notes.
Due:
Assignment
Due:
Thurs, 11/9/17 (stamped Mon, 11/13/17)
Finish "Empirical/Molecular Formulas" WS.
CHECK:
EF
1. C5H10O
2. KMnO4 (potassium permanganate)
3. AlCl3 (aluminum chloride)
4. BaSO4 (barium sulfate)
MF
1. C2H2 (dicarbon dihydride)
2. I2O5 (diiodine pentoxide)
3. C5H10O2
4. H2O2
5. C4H12N2
Due:
Assignment
Thurs, 11/2/17 (drilled Mon-Thurs, 11/6/17-11/9/17)
Practice "Mixed Naming and Formulas"... (25/day to finish by Thurs, 11/9)
Quiz on Thursday-Tuesday, 11/9/17, 11/13/17, 11/14/17
CHECK
1. lithium oxide; 2. difluorine monochloride; 3. sulfuric acid; 4. potassium sulfate; 5. tin (IV) sulfide; 6. cobalt (III) carbonate; 7. nitrogen monoxide; 8. uranium (IV) oxide; 9. nitric acid; 10. gold (III) nitrate; 11. aluminum sulfite; 12. cesium phosphide; 13. copper (II) hydroxide; 14. tetraphosphorus decoxide; 15. hydrochloric acid; 16. silver chloride; 17. magnesium chlorate; 18. calcium fluoride; 19. hexacarbon triselenide; 20. titanium (IV) phosphate; 21. tetraphosphorus octafluoride; 22. carbonic acid; 23. chromium (III) sulfide; 24. disulfur heptabromide; 25. zinc oxide; 26. K3PO4; 27. N2Br4; 28. H3PO4; 29. NaF; 31. K2HPO4; 32. H2SO3; 33. S3I; 34. SO3; 35. CoCl3; 36. Fe2(SO4)3; 37. Fe2S3; 38. Ca(ClO2)2; 39. HCN; 40. P2O5; 41. P5Cl3; 42. Li3N; 43. NO2; 44. ZnCl2; 45. MgCl2; 46. SO2; 47. Na2CO3; 48. CuF2; 49. (NH4)2S; 50. N3O2; 51. ZnO2; 52. Pb(NO3)4; 53. H2S; 54. AlBr3; 55. Ca(OH)2; 56. P3O8; 57. CuO; 58. CaCl2; 59. Al2O3; 60. Sn(ClO4)4; 61. HBr; 62. Ag2O; 63. SSe; 64. NaHCO3; 65. NH4I; 66. ZnI2; 67. HNO3; 68. FeSO4; 69. SiO2; 70. Mg3(PO3)2; 71. K3P; 72. KOH; 73. Co3(PO4)2; 74. HC2H3O2; 75. NiP; 76. Al2(CrO4)3; 77. (NH4)2Cr2O7; 78. aluminum oxide; 79. trisilicon pentoxide; 80. ammonium phosphate; 81. dinitrogen hexoxide; 82. hypochlorous acid; 83. magnesium iodide; 84. dinitrogen pentafluoride; 85. potassium bromide; 86. silicon dioxide; 87. triphosphorus nonabromide; 88. iron (II) iodide; 89. lithium hydrogen phosphate; 90. copper (I) oxide; 91. copper (II) carbonate; 92. oxalic acid; 93. silver nitrate; 94. magnesium nitride; 95. copper (II) bicarbonate; 96. dinitrogen monoxide; 97. tetraphosphorus octabromide; 98. gold (I) nitrite; 99. nickel (III) carbonate; 100. lead (II) hydroxide; 101. cobalt (III) sulfide; 102. ammonium bromide; 103. potassium permanganate; 104. cadmium sulfide
Due:
Per. 1, 3, and 6
Tues, 11/6/17 (stamped Wed, 11/7/17)
Finish "Moles and % Composition" WS.
Per. 2
Wed, 11/7/17 (stamped Thurs, 11/8/17)
Finish "Moles and % Composition" WS.
CHECK:
1. 14.1 mol C2H6
2. 0.5213 mol PbSO4
3. 8.3 L N2
4. 4.4 x 1021 f.un SnCl2
5. 69.5 g C10H22
6. 3.19 x 1023 molec CH3OH
7. 9.89 x 1022 atom Ne
8. 38.2 L HBr
9. 3.73 x 1023 molec CO2
10a. 11.2% H and 88.79% O
10b. 38.67% K, 13.86% N, and 47.47% O
10c. 38.76% Ca, 19.97% P, and 41.28% O
Due:
Fri, 11/3/17 (stamped Mon, 11/6/17)
Finish "Moles #2" WS.
Freon-12 should be 121 g/mol.
Due:
Assignment
Tues, 10/24/17 (drilled Mon-Fri, 10/30/17-11/3/17)
Make flashcards of your polyatomic ions' formulas and names.
Quiz on Friday, 11/3/17
Due:
Thurs, 11/2/17 (stamped Fri, 11/3/17)
Finish "Moles" WS.
Due:
Assignment
Wed, 11/1/17 (stamped Thurs, 11/2/17)
Finish "Naming and Formulas" WS.
CHECK:
Binary Ionic
sodium iodide; CuO; copper (I) chloride; BaO; iron (II) sulfide; AlCl3; magnesium sulfide; Fe2O3; lithium fluoride; PbO2; lead (II) sulfide; K2S; nickel (II) chloride; AgCl; tin (IV) chloride; CaBr2; cobalt (III) nitride; SrF2; zinc chloride; MnS
Binary Covalent
pentaphosphorus nonoxide; As2Br3; dinitrogen tetroxide; CCl4; phosphorus pentachloride; Si3N4; xenon tetrafluoride; Sb2O5; nitrogen triiodide; B2H6; sulfur dioxide; CO; diphosphorus tetraiodide; P4O10; chlorine dioxide; N2O3; tetraphosphorus trisulfide; CI4; dichlorine pentoxide; ClF3
Ternary Ionic
cadmium phosphate; Cr(OH)2; cadmium permanganate; Cr(CN)3; sodium acetate; NaHCO3; sodium nitrate; Al(NO3)3; barium sulfate; Co2(SO4)3; lead (IV) chromate; Mg(ClO3)2; potassium dichromate; (NH4)3PO4; cobalt (II) chromate; CuSO4; potassium permanganate; Al(ClO3)3; tin (II) phosphate; ZnCO3
Acids
nitric acid; H2SO4; nitrous acid; HCl; chromic acid; HC2H3O2; hydrobromic acid; H2CO3; hydrocyanic acid; HF; phosphoric acid; HClO3; permanganic acid; HClO; chlorous acid; HMnO4; perchloric acid; H2CrO4; sulfurous acid; HI
Due:
Assignment
Presentation standards
For your given category (e.g. covalent, acid, ionic w/ RN, ionic w/o RN, ionic -ide, ionic -ite/-ate), explain
Given the formula,
a) how to identify
b) how to name
Given the name,
c) how to identify
d) how to write the formula
| Eye Contact and Posture | Clarity and Volume | Content | Enthusiasm | Timing |
|
-refers to notes infrequently or not at all
-eye contact with entire audience
-hand placement, motions, and posture appropriate
|
-speaks clearly and distinctly
-uses consistent pacing
-volume is sufficient to be heard and understood throughout the room
-avoids filler words such as um, okay, like
-uses complete sentences and correct pronunciation
|
-uses correct subject vocabulary
-has a clear, concise introduction and conclusion
-demonstrates full understanding of topic
-no subject accuracy errors
|
-effectively uses inflection and pitch
-confident is evidence
|
-meets requirements of 4 minutes/group
|
Due:
Assignment
Fri-Mon, 10/26and30/17 HW (submitted Tues, 10/31/17 before 7:59 AM)
1. Compose your summary by hand or on a Google doc to save your work.
2. Copy-and-paste your summary here, choosing "Naming Ionic Compounds."
Due:
Assignment
Thurs, 10/26/17 HW (submitted Fri, 10/27/17 before 7:59 AM)
1. Compose your summary by hand or on a Google doc to save your work.
2. Copy-and-paste your summary here, choosing "Mass and Moles."
Due:
Assignment
Mon, 10/23/17 (stamped Tues, 10/24/17)
Finish "How to draw Lewis Dot Structure" backside.
Due:
Assignment
Fri, 10/20/17 (stamped Mon, 10/23/17)
Finish "Lewis Dot Structure and Polyatomic Ions" backside.
Due:
Assignment
10/13/17 HW (due in basket, Wed, 10/18/17)
Finish sky blue RS. Unit 3 - Atoms & Electrons
Test on Wednesday, 10/18/17
1. Historical evidence for the existence of atoms and the structure of the atom
-Dalton's Atomic Theory (5)
-describe the experiment that proved the existence of electrons
-describe the experiment that proved the existence of the nucleus
-draw a diagram of the Bohr model
-relative charge and approximate mass (amu) of the proton, neutron, and electron
2. Atoms are composed of protons, neutrons, and electrons
-identify the number of protons, neutrons, and electrons in a neutral atom
-difference between atomic number, mass number, and atomic mass
-define and illustrate an isotope
-calculate the average atomic mass given the abundance of each naturally occurring isotope
3. Electrons move about the atom in a cloud of quantized energy levels
-significance of flame tests in terms of energy level
-describe the electron cloud in terms of energy levels, sublevels, and orbitals
-draw an s-orbital, p-orbital, and a p-sublevel
-ground-state and core configuration
-identify the number of valence electrons
-draw electron dot structures for valence electrons
-octet rule for predicting the charge on an ion (s- and p-block elements)
"Light, Energy Levels, Electron Configuration" WS (optional):
1-8: red; violet; red; violet; red; violet; 550 nm; 550 nm
9-10. 5-->2; 2-->1
11. 1 sublevel; 2 sublevels; 3 sublevels; 4 sublevels
12. 2 e-; 6 e-; 10 e-; 14 e-
13. 8 e-
14. 32 e-
15. 1s 2s 2p 3s 3p 4s 3d 4p 5s 4d 5p 6s 4f 5d 6p 7s 5f 6d 7p
16. ends in 1s2, 2s2, 3d7, 4d10, 2p1, 3p6
17. Si, Ti, Sr, F, Ar, Tc
Due:
Assignment
Mon, 10/16/17 (stamped Tues, 10/17/17)
Finish "Electron Configurations 2" WS.
Due:
Assignment
Thurs, 10/12/17 (due in basket Fri, 10/13/17)
Sign progress report.
Also memorize filling order: 1s22s22p63s23p6etc.
Due:
Assignment
Tues, 10/10/17 (stamped Wed, 10/11/17)
Finish "Electron Cloud" WS.
Due:
Assignment
Fri, 10/6/17 HW (submitted Mon, 10/9/17 before 7:59 AM)
1. Compose your summary by hand or on a Google doc to save your work.
2. Copy-and-paste your summary here, choosing "Ions."
Due:
Assignment
Thurs, 10/5/17 HW (due in basket before the bell rings, Fri, 10/6/17)
Finish Lab - Isotopes Makes Cents.
Due:
Assignment
Wed, 10/4/17 (stamped Thurs, 10/5/17)
Finish "Average Atomic Mass" WS, both sides.
Check!
1. 39.09 amu
2. 14.01 amu (the typo should read "0.368%)
3. 35.45 amu
1. 107.87 amu
2. 55.85 amu
3. 47.88 amu
4. 195.1 amu
Due:
Assignment
Tues, 10/3/17 HW (submitted Wed, 10/4/17 before 7:59 AM)
1. Compose your summary by hand or on a Google doc to save your work.
2. Copy-and-paste your summary here, choosing "Average Atomic Mass."
Due:
Assignment
Mon, 10/2/17 HW (submitted Tues, 10/3/17 before 7:59 AM)
1. Compose your summary by hand or on a Google doc to save your work.
2. Copy-and-paste your summary here, choosing "Isotopes."
Due:
Assignment
Thurs-Fri, 9/28-29/17 ClassWork (CW) (submitted online before 7:59 AM Mon, 10/2/17 and hardcopy dropped in basket before bell rings on Monday)
2. Shared with [email protected]
3. Everyone prints a hard copy of their own document to be stapled to your data sheet
4. Everyone submits to turnitin.com
Turnitin.com codes per period:
Per. 1: 16163466
Per. 2: 16163478
Per. 3: 16163492
Per. 6: 16163495
Keycode for each period: chemishih
Due:
Assignment
Thurs, 9/28/17 (stamped Fri, 9/29/17)
Finish #1-3 for Mystery Powder.
Remember! Explain = Claim+Evidence+any relevant principle/theory
Due:
Assignment
9/26/17 HW (due in basket, Wed 9/27/17)
Finish royal blue RS. Unit 2 - Matter & Energy
Test on Wednesday, 9/27/17
1. There are three physical states of matter.
-distinguish between shape, volume, and compressibility
-describe phase changes
-distinguish between three states of matter in terms of strength of attraction between particles
2. Matter can be classified many ways.
-classify homogeneous or heterogeneous
-classify element, compound, or mixture
-distinguish between elements, compounds, and mixtures
-classify as metal, semimetal (metalloid), or nonmetal
-distinguish between metals, semimetals, and nonmetals on the basis of their characteristic properties
3. Matter undergoes many chemical and physical changes.
-classify chemical or physical property
-classify a chemical or physical change
-distinguish between elements, compounds, and mixtures in terms of separation via chemical or physical change
-experimental observations that provide evidence for chemical change
4. Chemical reactions obey the law of conservations of mass and energy.
-the mass sum of reactants equals the mass sum of products
-measure the specific heat of metal using calorimetry and the law of conservation of energy (e.g. +qsys = -qsurr; -exo = +endo)
Check:
"Reciew - Ch. 3 Matter and Energy"
1. liquid solid gas liquid liquid solid
2. freezing deposition melting boiling condensation sublimation
3. element mixture mixture compound mixture compound element mixture
4. het hom hom het het hom hom het
5. 2 aluminum 3 sulfur 9 oxygen; 1 magnesium 2 oxygen 2 hydrogen; 3 barium 2 phosphorus 8 oxygen; 1 calcium 2 nitrogen 6 oxygen
6. six carbon, eight hydrogen, seven oxygen; three carbon, six hydrogen, three oxygen
7. SO2 C10H16N5O13P3
8. P c c P c P P P
9. P c c P P c c P c P P c c c
10. sm M M nm M sm nm M nm
10. color ∆, temperature ∆, formation of precipitate or gas
11. 14.91 g; 0.055 g; 1500 kcal
12. evaporation, distillation
13. M M nm M nm nm nm M nm M
16. magnet; water; melting point; evaporation; distillation; time, maybe distillation
Due:
Assignment
Mon, 9/25/17 (stamped Tues, 9/26/17)
Finish "Review - Ch. 3" WS.
Due:
Assignment
Fri, 9/22/17 (stamped Mon, 9/25/17)
Finish "Definite Proportions and Conservation of Mass" WS.
Check!
1. 15 atoms total
2. C8H11NO3
3. 14 atoms total
4. C3H5O3(NO2)3
5. 1.600 g O2
6. 0.975 g ZnS
7. 0.252 g S
Due:
Assignment
Wed, 9/20/17 ClassWork (CW) (submitted online before 7:59 AM Fri, 9/22/17 and hardcopy dropped in basket before bell rings on Friday)
2. Shared with [email protected]
3. Everyone prints a hard copy of their own document to be stapled to your data sheet
4. Everyone submits to turnitin.com
Turnitin.com codes per period:
Per. 1: 16163466
Per. 2: 16163478
Per. 3: 16163492
Per. 6: 16163495
Keycode for each period: chemishih
Due:
Assignment
Thurs, 9/21/17 HW (submitted Fri, 9/22/17 before 7:59 AM)
1. Compose your summary by hand or on a Google doc to save your work.
2. Copy-and-paste your summary here, choosing "Fundamental Laws."
Due:
Assignment
Tues, 9/19/17 (stamped Wed, 9/20/17)
Finish "Physical vs. Chemical" WS.
Due:
Assignment
Wed-Thurs, 9/13-14/17 ClassWork (CW) (submitted online before 7:59 AM Tues, 9/19/17 and hardcopy dropped in basket before bell rings on Tuesday)
2. Shared with [email protected]
3. only ONE person prints a hard copy for basket
4. only ONE person submits to turnitin.com
Turnitin.com codes per period:
Per. 1: 16163466
Per. 2: 16163478
Per. 3: 16163492
Per. 6: 16163495
Keycode for each period: chemishih
Due:
Assignment
Fri, 9/15/17 HW (submitted Mon, 9/18/17 before 7:59 AM)
1. Compose your summary by hand or on a Google doc to save your work.
2. Copy-and-paste your summary here, choosing "Classification of Matter."
Due:
Assignment
Thurs, 9/14/17 HW (submitted Fri, 9/15/17 before 7:59 AM)
1. Compose your summary by hand or on a Google doc to save your work.
2. Copy-and-paste your summary here, choosing "Experimental Design."
Due:
Fri, 9/15/17 notes
Absent kiddos & first period, these slipped my mind. Please check your notes.
Due:
Assignment
9/11/17 HW (due in basket, Tues 9/12/17)
Finish violet RS. Unit 1 - Metric System
Test on Tuesday, 9/12/17
1. Conversion factors and Dimensional Analysis
- metric vs. English
- cubic units to other cubic units
- prefixes (nano, micro, milli, centi, deci, kilo)
- cubic units to liquid volume measure
2. Significant Figures
- how to make valid measurements
-counting sfs
-rounding rules (add/subtract vs. multiply/divide)
3. Units
-length, mass, volume, temperature, time
-common instruments to the above
4. Density
-how to calculate density
-how to use density as a CF to convert between mass and volume
-predicting sinking/floating
5. Heat vs. Temperature
-q, T, and C
- ºC <----> K
-Calculate one of the unknown given three of the following: q, m, C, or T
-directly and inversely proportional relationships
Due:
Assignment
Fri, 9/8/17 (stamped Mon, 9/11/17)
Finish "Heat and Temperature"
CHECK!
1a. 273K
1b. 400 K
1c. 200 K
1d. 0 K
2a. -273 ºC
2b. -200 ºC
2c. 200 ºC
2d. 0 ºC
3. 30000 J or 6000 cal
4. 67.5 cal
5. 56.5 g
6. 0.0308 cal/gºC
7. 59.2 ºC
8. 55 ºC
9. 1.1 J/gºC
10. 0.26 cal/gºC
11*. 34 ºC
Due:
Assignment
If you received a 5.5 or higher on your Measurement/Sf Lab, please correct your mistakes and re-submit on Monday into the basket. You may also have to prove to me that you can made valid measurements and count sfs.
Due:
Assignment
Thurs, 9/7/17 HW (due in basket before the bell rings, Fri, 9/8/17)
Finish Lab - Target Density.
Due:
Assignment
Fri, 9/1/17 (stamped Tues, 9/5/17)
Finish "q=mC∆T."
CHECK!
6a. 0.850 Cal
6b. 1800 J
7a. 1860 J
7b. 110 cal
8. 0.460 J/gºC
9. 3.0 x 102 J
10. 230 ºC
11. 1.8 J/gºC
12. 14000 J
13. 0.140 J/gºC
14. 32 ºC
15. 1.0 x 103 cal
16. 5440 J
17. 150 J
18. 5.0 x 102 J
19. 7000 J
Due:
Assignment
Thurs, 8/31/17 HW (submitted Fri, 9/1/17 before 7:59 AM)
1. Compose your summary by hand or on a Google doc to save your work.
2. Copy-and-paste your summary here, choosing "Calorimetry."
Due:
Assignment
Tues, 8/29/17 HW (due in basket before the bell rings, Wed, 8/30/17)
Finish Lab - Density.
Due:
Assignment
Mon, 8/28/17 (stamped Tues, 8/29/17)
Finish "Unit Analysis."
CHECK!
1a. 6.8 x 100 dollars
1b. 3.10 lb
2a. 115 in
2b. 648 mi if using the CF that has 3 sf
2c. 97.1 oz
2d. 1.7 x 104 mL
2e. 2.43 yd
3a. 15.92 €
3b. $126
4. 420 km
5. 29 m/s
6. 0.756 mg
Due:
Assignment
Fri, 8/25/17 (stamped Mon, 8/28/17)
Finish "Unit Analysis 2".
CHECK!
1. 0.236 g Al
2. 56.3 g H2O
3. 436 m2
4. 0.404 hectares
5. U.S. is cheaper (6.85 kr/L < 14.12 kr/L)
6. 2.11 £ (English pounds)
7. 244 in3
8. 1.36 x 104 kg/m3
9. 7720 or 7.72 x 103 tablets
10. a. 680 mi and 180 mi; b. difference of $50. <-- 2sf
"Just For Fun" but idk about the sfs... ~2.53 frizzles
Due:
Assignment
Wed-Thurs 8/23-24/17 HW (stamped Fri, 8/25/17)
Finish "Conversions" WS.
Finish "Density practice" WS.
CHECK "Conversions 2"
1. 9.23E11 nm
2. 4.851E-2 dg
3. 1.2 cups
4. 5.18E-2 mi
5. 3.13E6 cm3
6. 55.9 L
7. 1.66E9 g/m2
8. 3.1E-4 yd3/s2
Due:
Assignment
Tues, 8/22/17 HW (due in basket before the bell rings, Wed, 8/23/17)
Finish Lab - Measuring and Sfs.
Due:
Assignment
Mon, 8/21/17 HW (due Tues, 8/22/17)
Finish "Operations w/ Sfs."
Due:
Assignment
Fri, 8/18/17
Once in a Titan blue ;) moon, NO HW, so why this post?
WEEK 1 (TECH) FORGIVENESS
Please make sure you turn in all of Week 1 assignments either online via this GF by Monday, 8/21 7:59 AM or into my basket before lunch.
Chronologically:
1. (yellow) Syllabus, signed
2. (white) Safety contract, signed
3. (pink) Student info sheet, filled and written letter on back
4. POGIL Significance and Measurement summary
5. POGIL Significant Zeros summary
Due:
Assignment
Thurs, 8/17/17 HW (submitted Fri, 8/18/17 before 7:59 AM)
1. Compose your summary by hand or on a Google doc to save your work.
2. Copy-and-paste your summary here, choosing "Significant Zeros."
Due:
Assignment
Wed, 8/16/17 HW (submitted Thurs, 8/17/17 before 7:59 AM)
1. Compose your summary on a Google doc to save your work.
2. Copy-and-paste your summary here, choosing "Significant Digits and Measurements."
Due:
Assignment
Tues, 8/15 HW (always due next day, e.g. Wed, 8/16, unless stated otherwise)
1. Get syllabus (yellow) & safety contract (white) signed.
2. Finish writing Student Info Sheet (pink)
2. Get materials (writing instruments--colors are great, 2-in 3-ring binder--useful, but whatever your organizational preference, go for it) for class (P.S. scientific calculators are @Target $9!)